Draw The Lewis Structure For Sih4
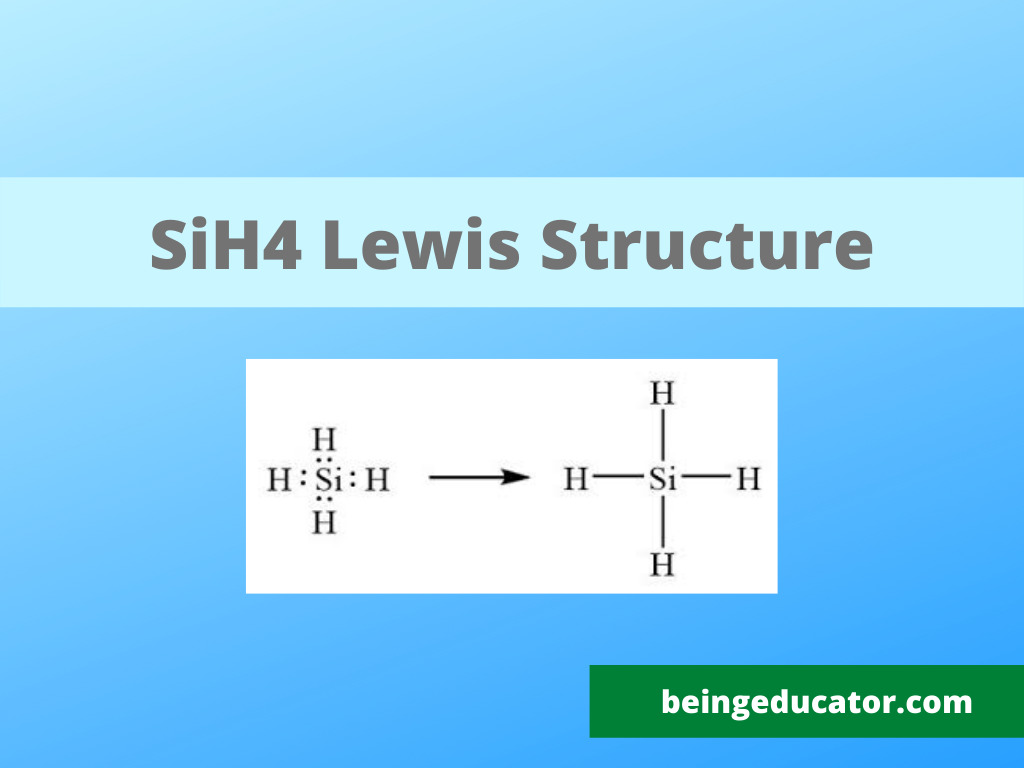
Draw The Lewis Structure For Sih4 - Here, the given molecule is sih4. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Draw the lewis symbol for each element in the molecule. Draw a lewis structure for silane (sih4) and predict its molecular geometry. Draw the lewis structure of sih4. Select draw rings more iii si h this problem has been solved! Draw the lewis structure for each molecule. Web to properly draw the sih 4 lewis structure, follow these steps: Sih4 is also called silane or monosilane, it is a colorless flammable and poisonous gas with a strong pungent odor. Each hydrogen atom should have two electrons around it, one from the bond and one as a lone pair. Web 6 steps to draw the lewis structure of sih4 step #1: While selecting the center atom, always put the least. Draw the lewis structure for co. A draw the lewis structure for sih4 in the window below and then decide if the molecule is polar or nonpolar. Draw the lewis structure for each molecule. Web best matched videos solved by our expert educators. Include lone pairs, as needed. Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. Draw the lewis structure for co. Draw a lewis structure for silane (sih4) and predict its molecular geometry. Web draw the lewis structure for sih4. Web chemistry learning made easy.this tutorial will help you deal with the lewis structure and moleculargeometry for silane (sih4). Welcome back to our channel, and in today’s video, we will help you do sih4 lewis structure. Here, the given molecule is sih4. Draw the lewis structure for each molecule. Okay, so you asked to draw the loose structure of i h four. Web draw the lewis structure for sih4. #1 draw a rough sketch of the structure first, determine the total number of valence electrons Web 6 steps to draw the lewis structure of sih4 step #1: Include lone pairs, as needed. Draw the lewis structure for co. Calculate the total number of valence electrons. Web to draw the lewis structure for sih4, follow these steps: Web h2co draw a lewis structure for the following three molecules. The sum of the valence electrons is. Draw the lewis structure for each molecule. Determine the total number of valence (outer shell) electrons in the molecule or ion. Predict the geometry of this compound. Web drawing lewis structures for molecules with one central atom: Web best matched videos solved by our expert educators. Sha s i h a. Web this problem has been solved! Lewis structure shows the bonding between atom of a molecule as well as the lone pairs of electrons… Count the number of electrons around each atom. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web draw the lewis structure for sih4. Web this problem has been solved! Sha s i h a. Draw the lewis structure for each molecule. #1 draw a rough sketch of the structure #2 next, indicate lone pairs on the atoms #3 indicate formal charges on the atoms, if necessary let’s break down each step in more detail. Include lone pairs, as needed. The sum of the valence electrons is. Is this compound ionic, polar covalent, or nonpolar covalent? Draw the lewis structure for co. Draw a lewis structure for silane (sih4) and predict its molecular geometry. Draw the lewis symbol for each element in the molecule. Count the number of electrons around each atom. For the sih4 structure use the periodic table to find the total number of valence electron.more. Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. H2co there are 4 steps to solve. Web let us determine the lewis structures of sih4 sih 4, cho−2 cho 2 −, no+ no +, and of2 of 2 as examples in following this procedure: The sum of the valence electrons is. Determine the total number of valence (outer shell) electrons in the molecule or ion. Web to draw the lewis structure for sih4, follow these steps: Each hydrogen atom should have two electrons around it, one from the bond and one as a lone pair. Web to properly draw the sih 4 lewis structure, follow these steps: Count the number of electrons around each atom. Calculate the total number of valence electrons. Is this compound ionic, polar covalent, or nonpolar covalent? #1 draw a rough sketch of the structure #2 next, indicate lone pairs on the atoms #3 indicate formal charges on the atoms, if necessary let’s break down each step in more detail. Web the lewis structure for sih 4 has 8 valence electrons available to work with. Find the total valence electrons in sih4. Welcome back to our channel, and in today’s video, we will help you do sih4 lewis structure. Draw the lewis structure of sih4. In order to draw the lewis. Web we have got our most suitable lewis structure sketch for sih4.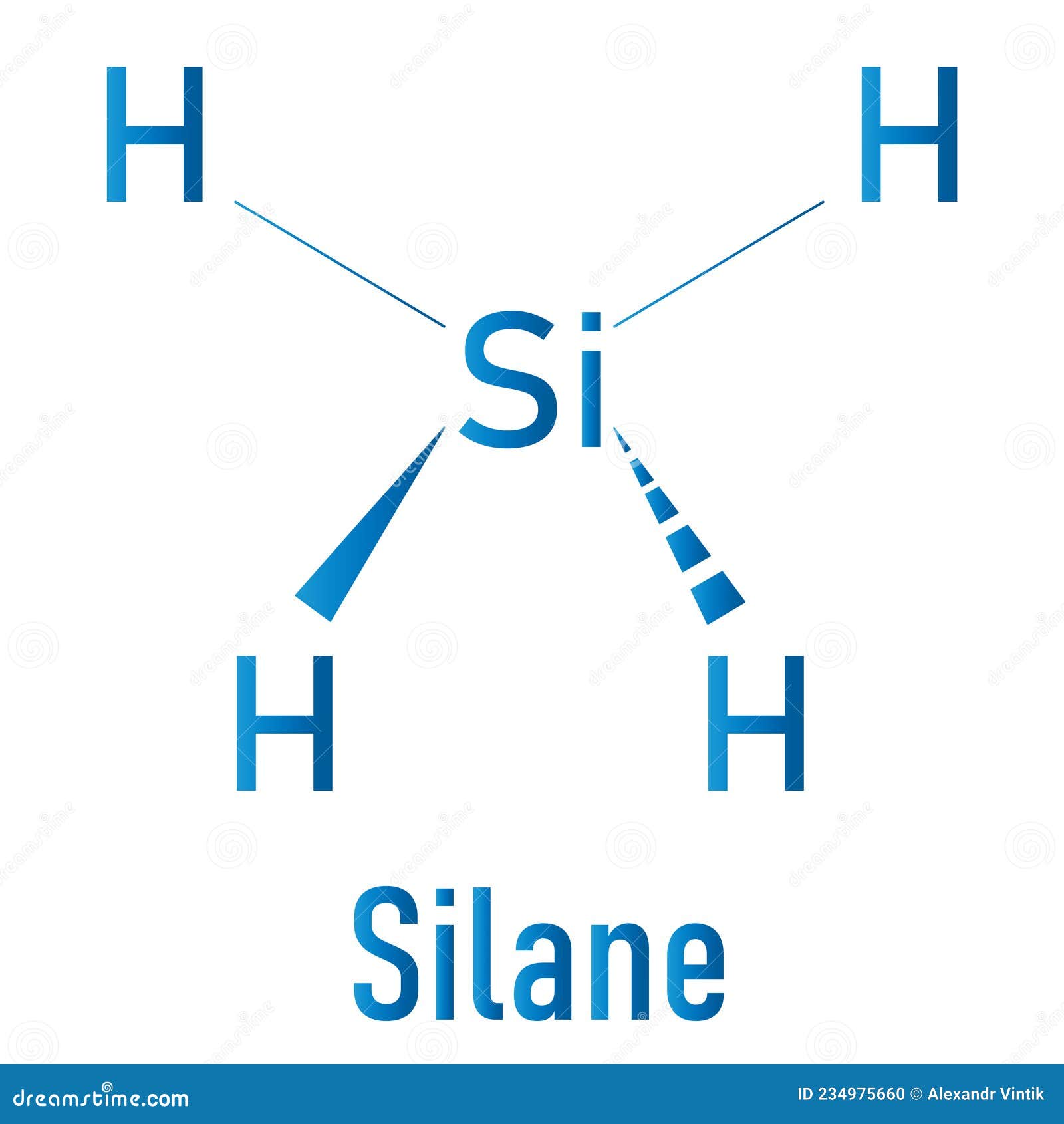
Silane SiH4 Molecule. Skeletal Formula Stock Vector Illustration of

diagrama de lewis del enlace siH4 Brainly.lat
Solved a Draw the Lewis structure for SiH4 in the window

SiH4 Lewis Structure (Silicon Tetrahydride) YouTube
[Solved] Draw the Lewis structure for SiH 4 in the window below and
Solved a Draw the Lewis structure for SiH4 in the window

Is SiH4 Polar or Nonpolar? Techiescientist
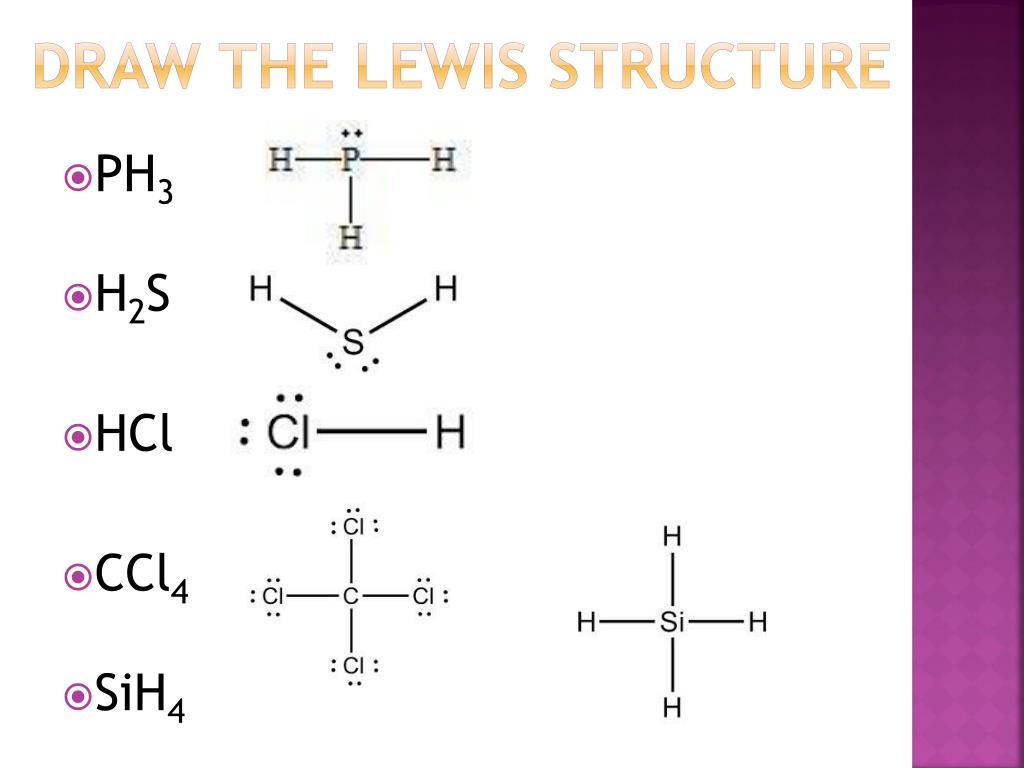
PPT Covalent Bonds PowerPoint Presentation, free download ID3048466

SiH4 Molecular Geometry, Bond Angles (and Electron Geometry) YouTube
Lewis Structure, Hybridization, Polarity and Molecular Geometry of SiH4
Draw The Lewis Structure For Each Molecule.
Web H2Co Draw A Lewis Structure For The Following Three Molecules.
For A Molecule, We Add The Number Of Valence Electrons On Each Atom In The Molecule:
Here, The Given Molecule Is Sih4.
Related Post: