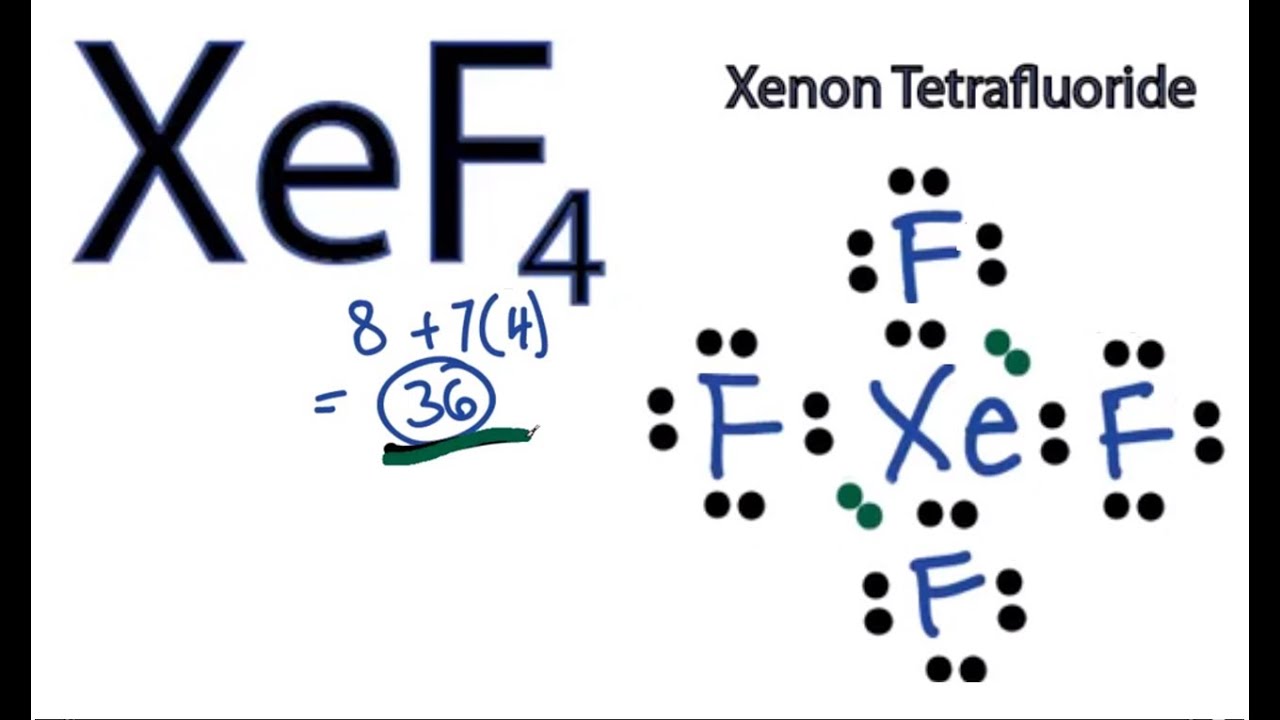
Draw The Lewis Structure For Xef2
Draw The Lewis Structure For Xef2 - The central xenon atom a. Web watch on steps of drawing xef2 lewis structure step 1: Xenon (xe) is in group 18 of the periodic table and has 8 valence electrons. It is bonded to 2 atoms of fluorine (f), one on each side of the molecule. С p opy aste []* с chemdoodle is xef2 polar or nonpolar? This problem has been solved! Determine the total number of valence electrons in xef2 by adding the valence electrons of each atom. Knowing how many valence electrons there a. I quickly take you through how to draw the lewis structure of xef2 (xenon difluoride). Xef2 lewis structure involves 1 atom of xenon and 2 atoms of fluorine. I quickly take you through how to draw the lewis structure of xef2 (xenon difluoride). Knowing how many valence electrons there a. Since we have two fluorine atoms, the total number of valence electrons is 8 (xe) + 2 (7) (f) = 22. Xenon is an inert gas element. Steps for drawing lewis structure of xef2. D) what are the bond angles? Determine the total number of valence electrons: Find the total valence electrons in xef2 molecule in order to find the total valence electrons in xef2 (xenon difluoride) molecule, first of all you should know the valence electrons present in xenon atom as well as fluorine atom. If there are more electrons than it, then. For the xef2 structure use the periodic table to find the total number of valence electron. Web drawing the lewis structure for xef 2. Web xef2 lewis structure is the abbreviation of xenon difluoride. Web to draw the xef2 lewis structure, follow these steps: Web chemistry chemistry questions and answers draw a lewis structure for xef2 and answer the following. Web lewis dot structure of xef2 (xenon difluoride) 97,881 views. In the ammonia molecule a lone pair on nitrogen resides in a (n) a. Ii the number of lone pairs the number of single bonds the number of double bonds = 2. For the central xenon atom: Web 5 steps to draw the lewis structure of xef2 step #1: Web © 2023 google llc for the xef2 structure use the periodic table to find the total number of valence electrons for the xef2 molecule. Web by using the following steps, you can easily draw the lewis structure of xef 2: Web watch on steps of drawing xef2 lewis structure step 1: #1 first draw a rough sketch first, determine. Draw the lewis structure for xef2 a) how many groups (atoms and lone pairs) surround the central oxygen? C) what is the molecular shape of this molecule? Determine the total number of valence electrons in xef2 by adding the valence electrons of each atom. Hydrogen (h) only needs two valence electrons to have a full outer shell. D) what are. Web science chemistry chemistry questions and answers draw the lewis structure for xef2 in the window below and then answer the questions that follow. Lewis diagram of xenon difluoride (xef₂) in some molecules, the central atom exceeds the octet rule (is surrounded by more than eight electrons). If there are more electrons than it, then that compound donates the electron.. #1 first draw a rough sketch #2 mark lone pairs on the atoms #3 calculate and mark formal charges on the atoms, if required let’s discuss each step in more detail. For the xef2 structure use the periodic table to find the total number of valence electron. C) what is the molecular shape of this molecule? Web to draw the. We use dots to represent outer shell electrons and lines to represent the bond type. Web to draw the xef2 lewis structure, follow these steps: Xef2 lewis structure involves 1 atom of xenon and 2 atoms of fluorine. If there are more electrons than it, then that compound donates the electron. Draw the lewis structure for xef2 a) how many. C) what is the molecular shape of this molecule? It is one of those rare compounds which involve noble gases despite their strong stability. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web chemistry chemistry questions and answers draw a lewis structure for xef2 and answer the following questions based on your. This problem has been solved! Web a video explanation of how to draw the lewis dot structure for xenon difluoride, along with information about the compound including formal charges, polarity. Web 5 steps to draw the lewis structure of xef2 step #1: It is bonded to 2 atoms of fluorine (f), one on each side of the molecule. C) what is the molecular shape of this molecule? You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web drawing the lewis structure for xef 2. Ii the number of lone pairs the number of single bonds the number of double bonds = 2. Web watch on steps of drawing xef2 lewis structure step 1: For the xef2 structure use the periodic table to find the total number of valence electron. The central xenon atom a. What is the electronic geometry of this molecule (look at atoms and lone pairs)? As we know xenon lies in. There are a total of 5 electron pairs in this lewis structure. This rule states that every molecule should have eight electrons in its outer shell of an atom to be stable. For the central xenon atom:
Xef2 Lewis Structure Lone Pairs Drawing Easy

XeF2 Lewis Structure How to Draw the Lewis Structure for XeF2 YouTube

Hello Guys! Today we are going to look at the Lewis Structure of XeF2
36+ Xef2 Lewis Structure Molecular Geometry Image GM
[Solved] Identify the correct Lewis structure for XeF2. O F=Xe=F O
![Molecular geometry of XeF2 [with video and free study guide]](https://aceorganicchem.com/chemistry/wp-content/uploads/2023/06/XeF2-lewis.jpg)
Molecular geometry of XeF2 [with video and free study guide]

XeF2 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram

The Lewis Structure Of Xef2 Understanding The Bonding Of Xenon

The Lewis Structure Of Xef2 Understanding The Bonding Of Xenon

XeF2 Lewis Structure How to Draw the Lewis Structure for XeF2 (Xenon
Obeys The Octet Rule B.
Lewis Diagram Of Xenon Difluoride (Xef₂) In Some Molecules, The Central Atom Exceeds The Octet Rule (Is Surrounded By More Than Eight Electrons).
#1 Draw Skeleton #2 Show Chemical Bond #3 Mark Lone Pairs #4 Calculate Formal Charge And Check Stability (If Octet Is Already Completed On Central Atom) Let’s One By One Discuss Each Step In Detail.
Web The Lewis Structure Of Xenon Difluoride (Xef2) Consists Of A Xenon (Xe) Atom At The Center.
Related Post: