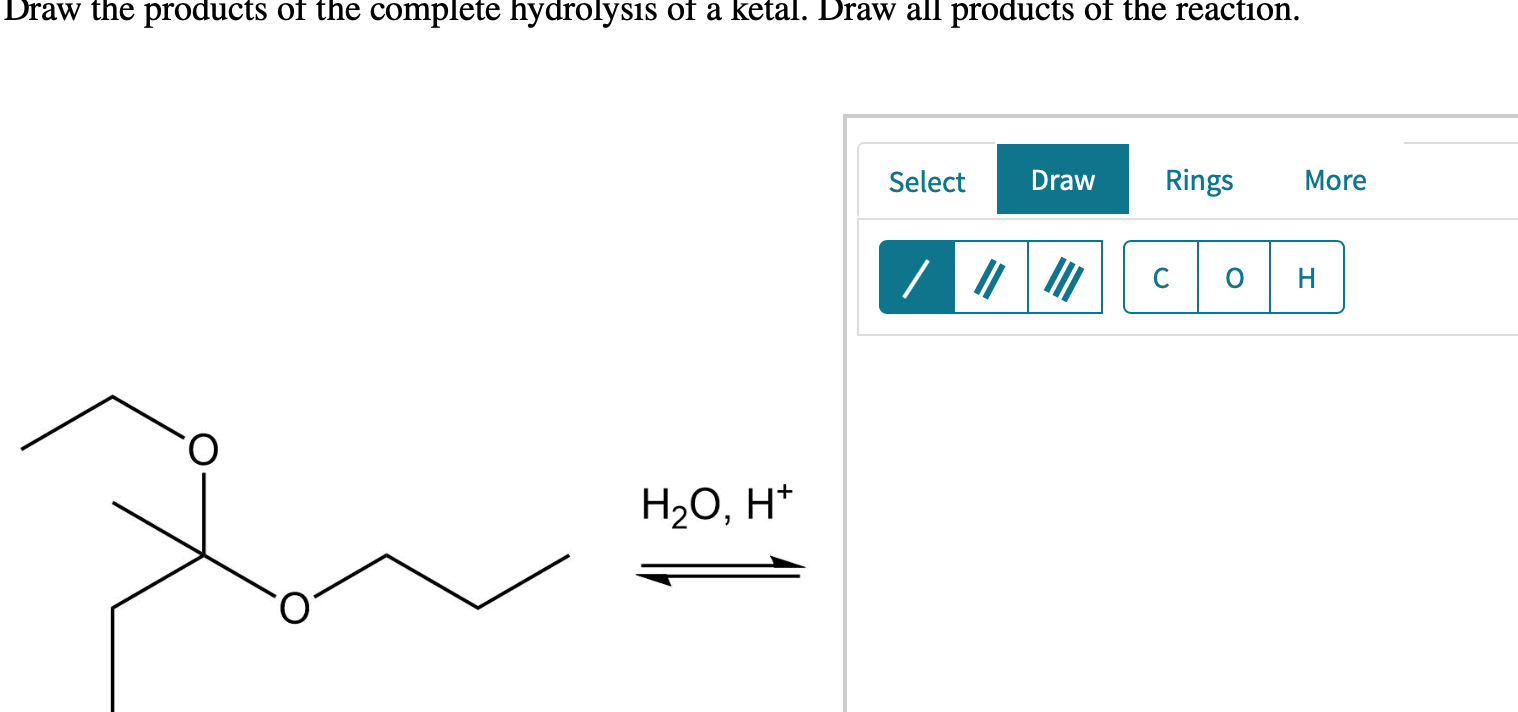
Draw The Products Of The Hydrolysis Shown.
Draw The Products Of The Hydrolysis Shown. - Web sal said that hydrolysis is using water to break something down, but in his diagram, there are the two glucose molecules and a water molecule (the h₂o below the two). The single bond is active by default. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Select draw 3 → rings more c 0 h frase 2 q expert solution trending now this is a popular solution! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. This results in the formation of a carboxylic acid (rcooh) and an alcohol (roh). Draw the carboxylic acid product of the acid hydrolysis of methanamide, shown here. In your answer, you should use appropriate technical terms, spelt correctly. Therefore, the products of the hydrolysis shown would be a carboxylic acid and an alcohol. Web exercise 15.8.1 15.8. Web sal said that hydrolysis is using water to break something down, but in his diagram, there are the two glucose molecules and a water molecule (the h₂o below the two). Would that mean that if two glucose molecules bonded together wouldn't they just break themselves apart because of the water molecules that is there? Web draw the products formed. Web in the hydrolysis reaction shown here, the dipeptide is broken down to form two ionized amino acids with the addition of a water molecule. Draw the product of the attached reaction: Web science chemistry draw the products of the given hydrolysis reactions. Draw the molecule on the canvas by choosing buttons from the tools (for bonds). You'll get a. In your answer, you should use appropriate technical terms, spelt correctly. Would that mean that if two glucose molecules bonded together wouldn't they just break themselves apart because of the water molecules that is there? You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Because soaps are prepared by the alkaline hydrolysis. Web. Draw the products of attached reaction The hydrolysis of h2co3 (carbonic acid) would result in the formation of h2o (water) and co2 (carbon dioxide). Select pin erase ch3 this problem has been solved! Draw the products of the hydrolysis shown. ( ch3 h₂o+ select dig h draw the products of the given hydrolysis reactions. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Draw the products of attached reaction Web draw the products of the acetal hydrolysis reaction shown below. Acetal are the products of nuelophillic addition reaction of aldehyde and and alcohol to form cyclic… q: This is where we draw all of the product of. General, organic, and biochemistry 9th edition isbn: Hydrolysis reaction of esters produces the respective carboxylic acids and alcohols. Because soaps are prepared by the alkaline hydrolysis. The hydrolysis of h2co3 (carbonic acid) would result in the formation of h2o (water) and co2 (carbon dioxide). Therefore, the products of the hydrolysis shown would be a carboxylic acid and an alcohol. Web science chemistry draw the products of the given hydrolysis reactions. Web therefore, the products of the hydrolysis are methanol and hydrochloric acid. The hydrolysis of ch3 (methyl group) is not possible as it is not a complete molecule. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Include hydrogen atoms in both. Web therefore, the products of the hydrolysis are methanol and hydrochloric acid. Web next, we need to break the ester bond through hydrolysis, which involves the addition of water (h2o) to the ester bond. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web in the hydrolysis reaction shown here, the dipeptide is. In your answer, you should use appropriate technical terms, spelt correctly. When a base (such as sodium hydroxide [naoh] or potassium hydroxide [koh]) is used to hydrolyze an ester, the products are a carboxylate salt and an alcohol. The balanced chemical equation is: The hydrolysis of ch3 (methyl group) is not possible as it is not a complete molecule. You'll. When a base (such as sodium hydroxide [naoh] or potassium hydroxide [koh]) is used to hydrolyze an ester, the products are a carboxylate salt and an alcohol. Include hydrogen atoms in both structures. Would that mean that if two glucose molecules bonded together wouldn't they just break themselves apart because of the water molecules that is there? In typical reactions,. Web therefore, the products of the hydrolysis are methanol and hydrochloric acid. Draw the products of attached reaction This results in the formation of a carboxylic acid (rcooh) and an alcohol (roh). The product of the reaction is drawn here. Esters are neutral compounds, unlike the acids from which they are formed. Would that mean that if two glucose molecules bonded together wouldn't they just break themselves apart because of the water molecules that is there? The balanced chemical equation is: The hydrolysis of h2co3 (carbonic acid) would result in the formation of h2o (water) and co2 (carbon dioxide). Hydrolysis reaction of esters produces the respective carboxylic acids and alcohols. We have not questioned that exact. Draw a quantitative molecular picture that shows what this system looks like after the reactants are mixed in one of the boxes and the system reaches equilibrium. Web draw the skeletal formula of the organic product formed from the complete hydrolysis of pga by naoh(aq). Web chemistry chemistry questions and answers draw the products of the hydrolysis. Because soaps are prepared by the alkaline hydrolysis. Write an equation for the acidic hydrolysis of methyl butanoate and name the products. The hydrolysis of ch3 (methyl group) is not possible as it is not a complete molecule.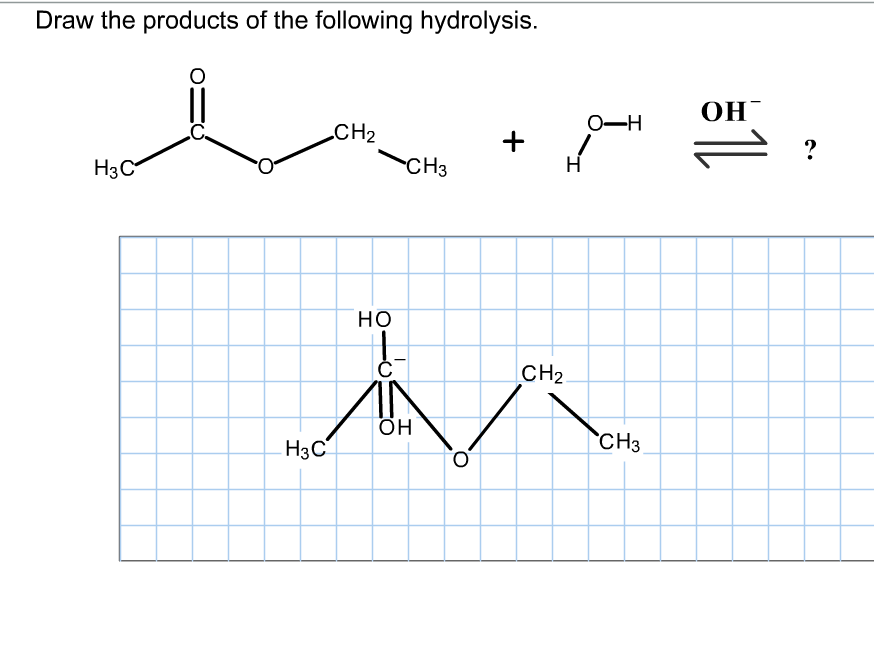
Chemistry Archive October 16, 2015
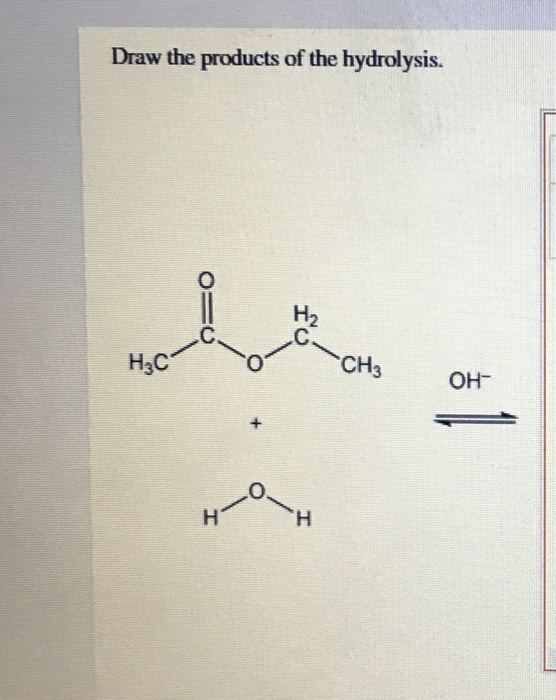
Solved Draw the products of the hydrolysis. НАС CH₃ OH
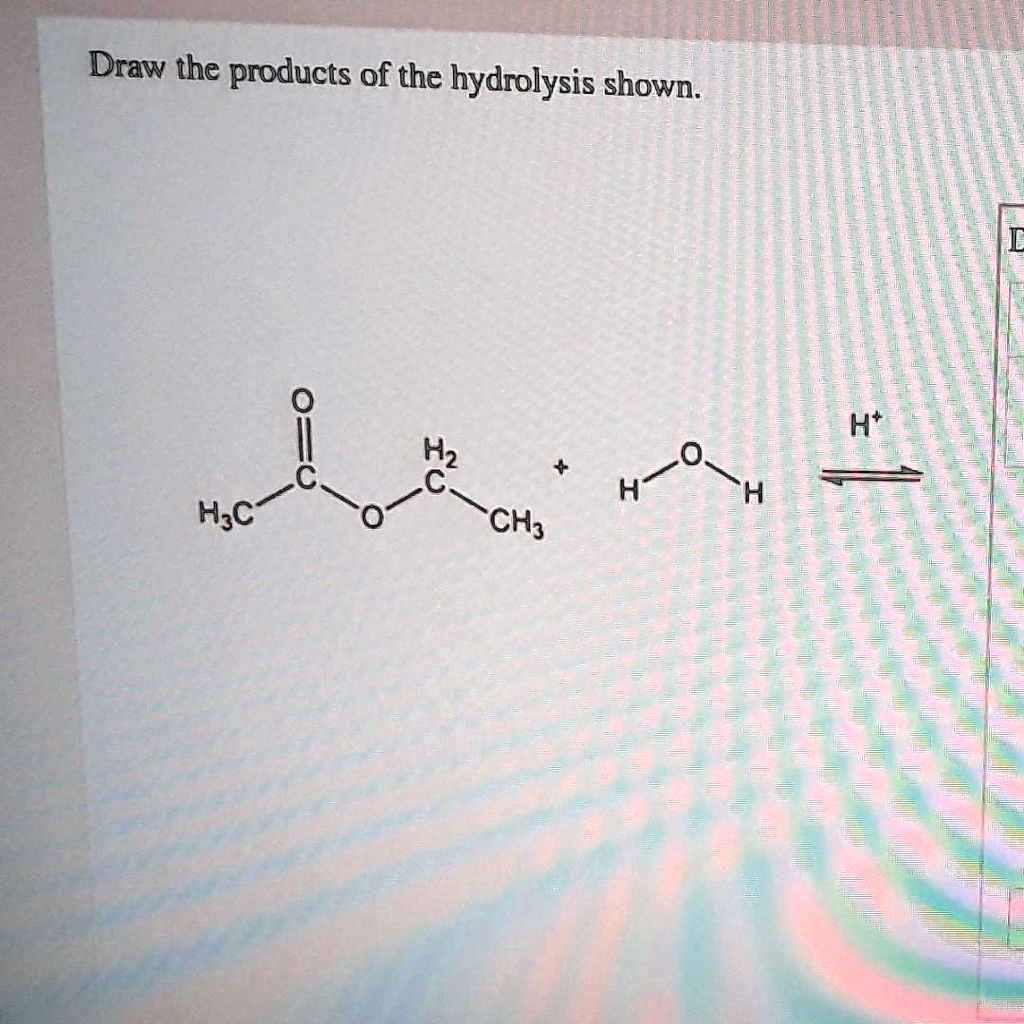
SOLVED Draw the products of the hydrolysis shown I Ht H2 HF H;c 'CH;
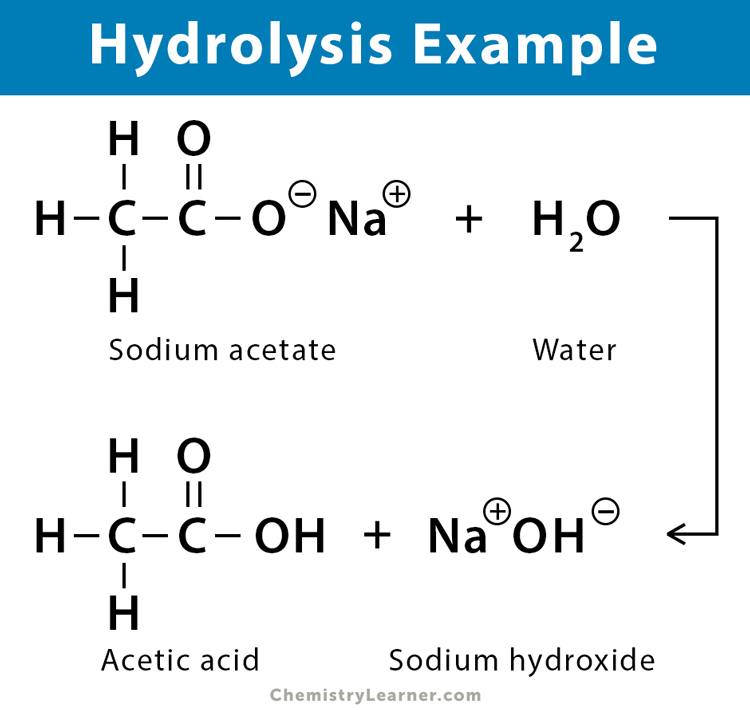
Hydrolysis Reaction Definition, Equation, and Applications
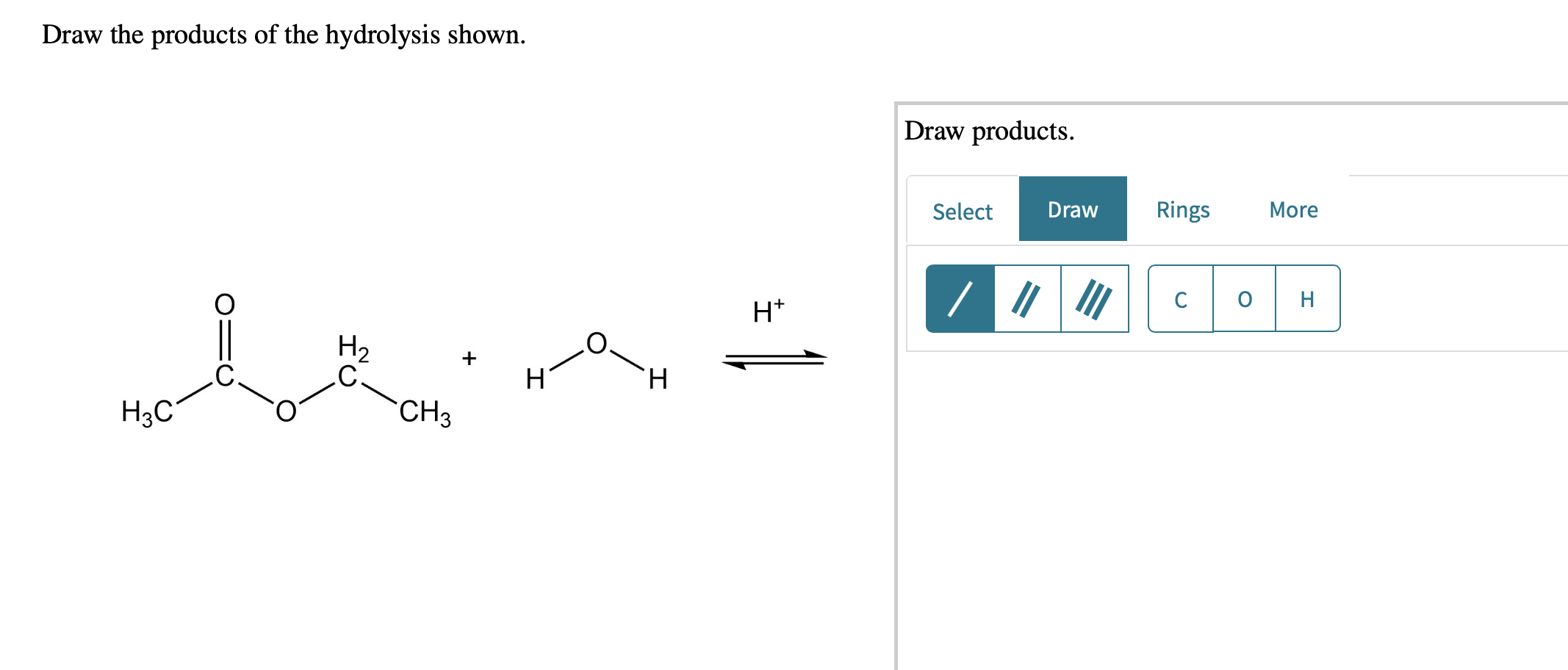
Solved Draw the products of the hydrolysis shown. Draw
[Solved] Draw the products of the hydrolysis shown. Draw products
[Solved] Draw the products of the hydrolysis shown. Draw products
Solved Draw the products of the complete hydrolysis of a
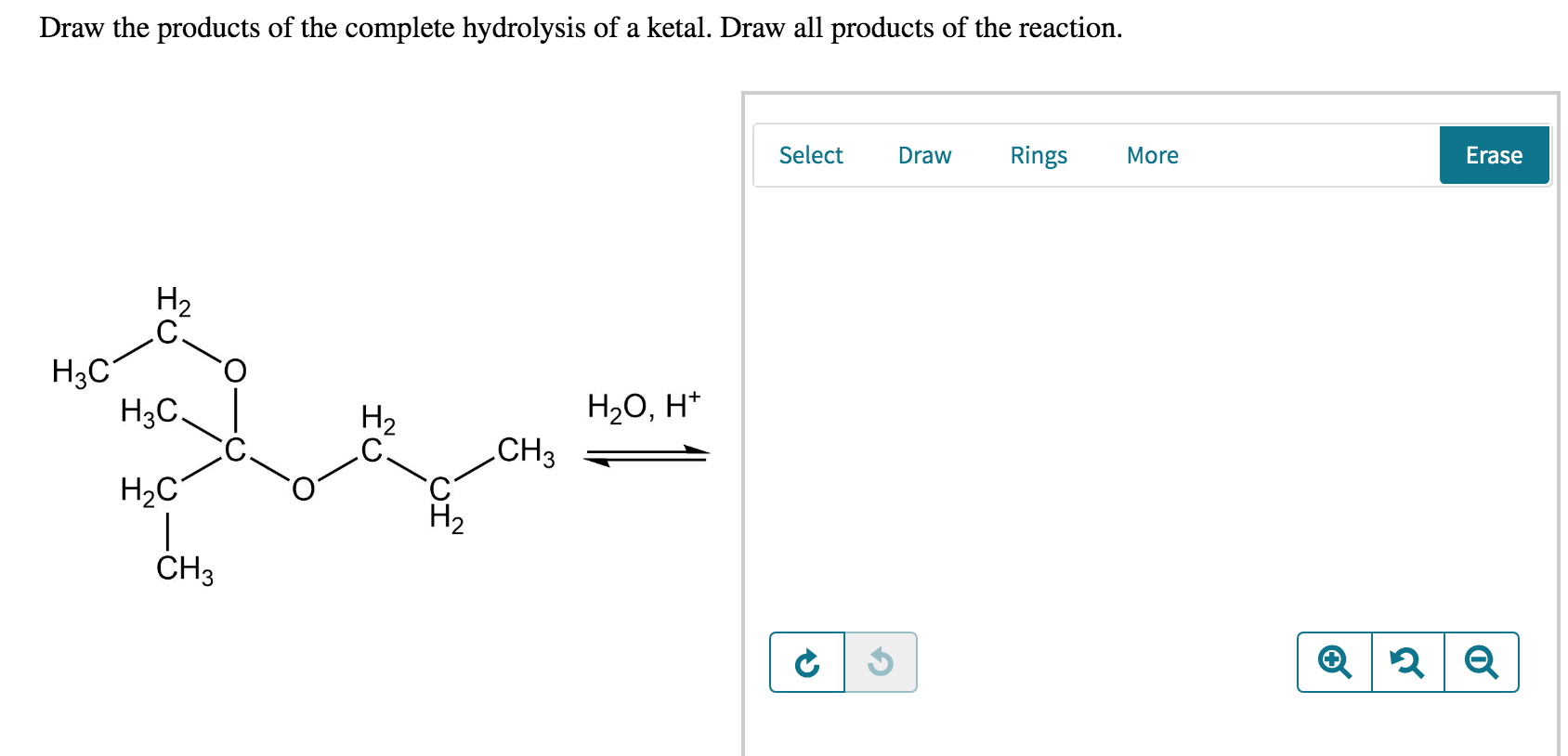
Solved Draw the products of the complete hydrolysis of a
:max_bytes(150000):strip_icc()/what-is-hydrolysis-375589-v2-5bdb527846e0fb002d706279.png)
An Explanation of the Process Hydrolysis
One Amino Acid Gets An Oxygen Atom And A Negative Charge, The Other Amino Acid Gets Two Hydrogen Atoms And A Positive Charge.
You'll Get A Detailed Solution From A Subject Matter Expert That Helps You Learn Core Concepts.
Web Science Chemistry Draw The Products Of The Given Hydrolysis Reactions.
Therefore, The Products Of The Hydrolysis Shown Would Be A Carboxylic Acid And An Alcohol.
Related Post: